Need for components of dental materials to be listed
There is an essential need for a consumer-based listing for components of dental materials including fillings, crowns, bridges, and other devices and implants. Less than thirty years ago, most food packaging did not include a list of ingredients and nutritional facts. However, consumers and other groups demanded this information be disclosed, and in 1990, food labeling became a requirement by the United States Food and Drug Administration (FDA). Unfortunately, to date, no such packaging list of the components of dental materials has been created for patients.
What are some of the components of dental materials including fillings, crowns, bridges?
Dental amalgams (silver) fillings, contain about 50% mercury, which is a known neurotoxin. This mercury is mixed with copper, silver, and tin. These fillings can also contain zinc and other metals, with at least one study finding the presence of lead and cadmium.
Other filling materials also contain components of which the average person is probably unaware. Composite fillings can contain bisphenol-A and fluoride, among other controversial components. Dental bridges, crowns, partial dentures, and implants can contain aluminum, chromium, cobalt, copper, gallium, gold, indium, iridium, iron, manganese, nickel, palladium, platinum, silver, titanium, vanadium and more. Gold restorations can also contain copper, gallium, indium, iridium, palladium, nickel, silver, tin, titanium, and zinc, as well as beryllium.
Safety Data Sheets: a worker-based listing of components of dental materials
The United States Department of Labor’s Occupational Safety and Health Administration (OSHA) requires manufacturers to produce safety data sheets (SDS, formerly known as material safety data sheets, or MSDS). The purpose of safety data sheets is to protect workers by supplying them with the most crucial facts about the hazardous materials at their jobsite, such as the physical properties of the material, proper storage and handling techniques, known health risks, and essential emergency procedures.
For example, manufacturers of amalgam fillings must create these information sheets for workers. Excerpts from the SDS for dental amalgam include compelling evidence about the known dangers of using mercury in fillings, such as this one:
- SDI Permite/Lojic +; Australia, Brazil, Ireland, and the USA: Toxicological Information/Chronic Health Effects: “Inhalation of mercury vapours, dusts or organic vapours, or skin absorption or mercury over long periods can cause mercurialism. Symptoms include tremors, inflammation of mouth and gums, excessive salivation, stomatitis, blue lines on gums, pain and numbness in extremities, weight loss, mental depression, and nervousness. Exposure may aggravate kidney disorders, chronic respiratory disease and nervous system disorders. May cause damage to blood, kidneys, liver, brain, peripheral nervous system, central nervous system.”
While this type of information would be very helpful to consumers, it is very rare that dental patients are presented with these safety data sheets. Additionally, the sheets only address the components of the particular dental material and not how these components might react with other dental materials in the mouth of the patient.
Mixing metals: crucial in understanding the components of dental materials
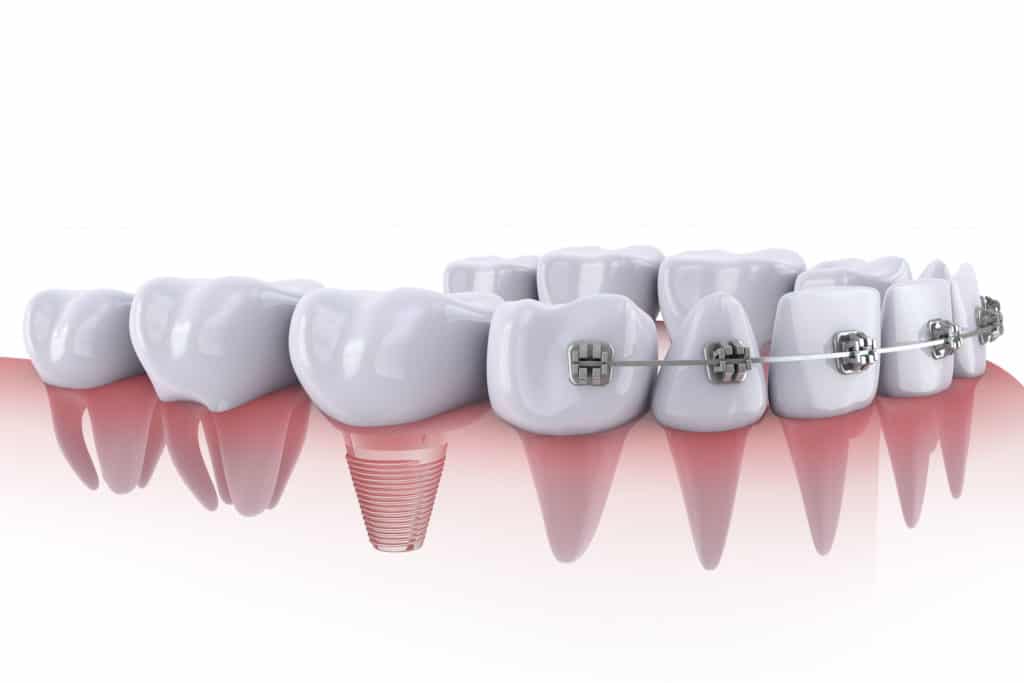
Mixtures of metals in different dental materials,such as fillings, crowns, bridges, and braces, can produce electric currents in the mouth.
A mouth with any amount of metallic dental materials has all the components needed to produce chemical reactions in a phenomenon known as oral galvanism, which simply means electric currents produced by chemical action in the mouth. Dental textbooks have included reference to oral galvanism for decades. Therefore, most dental authorities acknowledge oral galvanism, and Health Canada has even stated that “new amalgam fillings should not be placed in contact with existing metal devices in the mouth, such as braces.” However, knowledge of the existence of oral galvanism does not appear to have significantly altered most dental practices.
Conclusion about components of dental materials, including fillings, crowns, bridges
The number of dental alloys used since the 1980’s has noticeably increased, and more and more patients are being exposed to different dental materials, often with no practical knowledge about the potential hazards of the components of these dental materials. Meanwhile, research continues to present evidence of human health risks caused by mercury fillings, as well as other dental materials in the mouth.
The IAOMT works to educate dental and medical professionals and patients about biocompatibility and components of dental materials. Biocompatibility involves the use of dental materials that are best suited for the patients based on safety and their personal healthcare needs. Also, when dental mercury fillings are removed, the IAOMT encourages professionals and patients to ensure that measures are taken to mitigate mercury exposure by applying the IAOMT’s Safe Mercury Amalgam Removal Technique (SMART). Furthermore, the IAOMT researches alternatives to amalgam fillings and suggests that patients take an active role in making sure that they understand the components of dental materials being used in their mouths.